Evolution of Data Management in Clinical Research: Clinsight and CDISC.
30-11-2015 We have moved from paper data collection to the use of electronic tools, since both completing case report forms and the logistics underlying this type of support were cumbersome.
The introduction and use of said electronic tools have resulted in higher efficiency in terms of times as well as quality, traceability and integrity of all the data collected during a study. That is why, the area of Data Management is the one that has most drastically evolved in recent years.
In terms of time frames, paper studies require an additional investment to gather all the sheets of paper, send/receive documents by/to the CRO/sponsor, entering data into a database by a Data Entry, validation, query submission, resolution by the investigator, sending/receiving the query back to the CRO/sponsor, and correction of the database. All this extends the entire procedure of validation and requires the involvement of third parties to take care of the logistics aspect of sending/picking up the paper documents.
Some advantages of using electronic tools vs. paper include the cost-benefit ratio: paper documents require a physical space for filing, special requirements regarding safety and access control to said documents using fireproof file cabinets with combination/key locks, access control to these cabinets, etc. This also limits the logical way of access, i.e., you cannot look up data if you are not physically in the same place where the data are located. Electronic tools enable us to have much simpler storage, complete traceability regarding the person responsible for creating, changing, reviewing, or validating each data, and virtually unlimited online accessibility using a convenient system of permits per user, identifying each user with their own password-protected access.
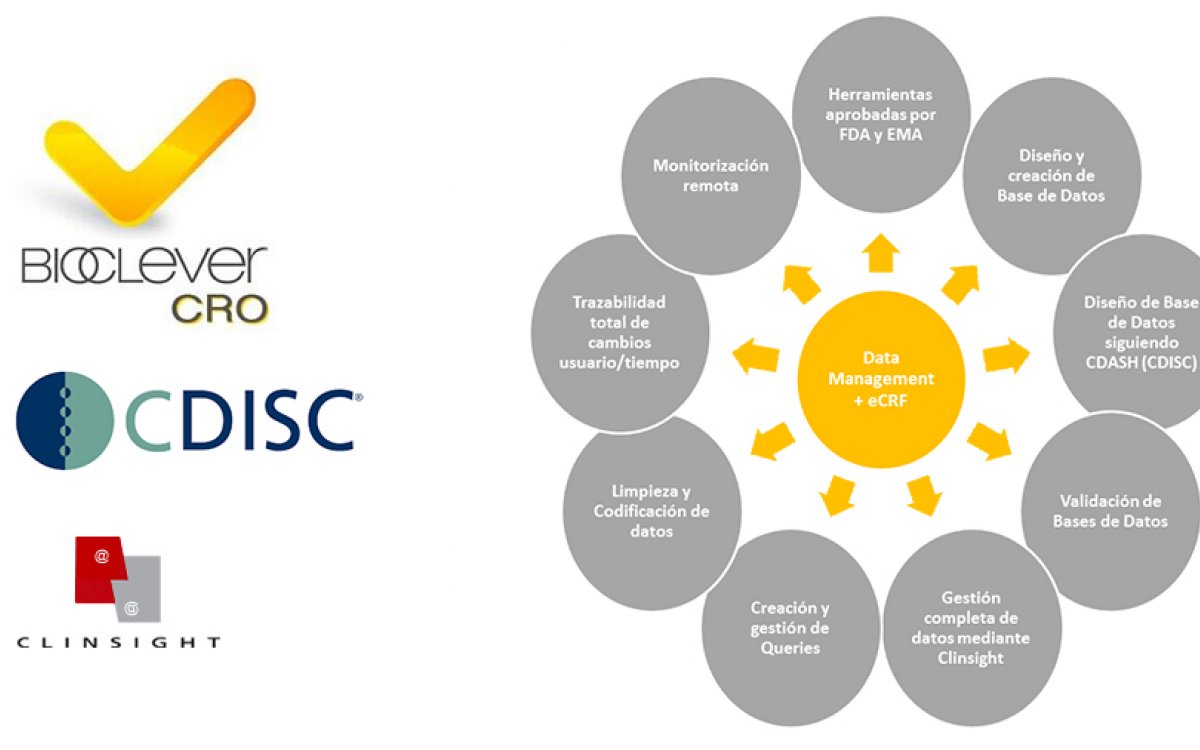
At BioClever, we have been working for years with one of the most powerful tools in the market: Clinsight, which complies with 21 CFR part 11 of the FDA and EMA standards and is certified by ISO 9001:2015. This is a highly versatile, intuitive tool with absolute traceability that allows to remotely monitor as well as manage a study, increasing the quality of data collection and review, and providing updated information on the status of the study at all times, which translates into a more efficient, transparent and reliable process with completely traceable and complete data. It is also one of the few tools that allow to export databases directly to SAS without having to export/import in Excel or another format.
Clinsight allows us to work with maximum accuracy, have each study with its own, much more personalized design and characteristics adapted to the client, but with the added value provided by a tool developed by a company of international renown that has been approved and validated by the main regulatory agencies. It offers a global solution for the development of a study, such as IWRS or SAE reconciliation, and allows to send SAEs at the same time as an investigator reports them. There is an increasing demand for these tools, highlighting the imperative need for even stricter data management processes; that is why, at BioClever, our entire Data Management team has been trained and certified to work pursuant to the guidelines of the CDISC (Clinical Data Interchange Standards Consortium).
Thus, using CDISC, we can obtain large databases in which we can connect several studies or trials to each other, which also helps us to perform new inter-study data analyses or search new observations that may not have been the original study objective. By using the CDISC methodology, you can standardize formats, both during the design of the database and for any subsequent statistical analyses.
In conclusion, we would like to highlight that the software that BioClever uses is 100% compliant with these standards; that is why, BioClever is currently and seamlessly developing its databases with CDISC for any studies that request it.
We appreciate you taking the time to read this article and hope it was of your interest; however, should you have any questions or comments you would like to share with us, please feel free to contact our professionals.
The introduction and use of said electronic tools have resulted in higher efficiency in terms of times as well as quality, traceability and integrity of all the data collected during a study. That is why, the area of Data Management is the one that has most drastically evolved in recent years.
Why is it best to use electronic tools than the traditional paper format?
The last 10 years have witnessed a true revolution in this area. Using paper as the main format for data collection is no doubt completely valid and still relevant today; however, in the past, the whole process, which included printing and distribution, was difficult and slow; report forms had mistakes; and there was an imperative need for exhaustive traceability as well as reducing times. All of this has contributed to limiting the use of paper to a few studies in which it remains the most efficient method because of its simplicity, specific study patient characteristics, or technical restrictions due to, among other things, the location where data will be collected.In terms of time frames, paper studies require an additional investment to gather all the sheets of paper, send/receive documents by/to the CRO/sponsor, entering data into a database by a Data Entry, validation, query submission, resolution by the investigator, sending/receiving the query back to the CRO/sponsor, and correction of the database. All this extends the entire procedure of validation and requires the involvement of third parties to take care of the logistics aspect of sending/picking up the paper documents.
Some advantages of using electronic tools vs. paper include the cost-benefit ratio: paper documents require a physical space for filing, special requirements regarding safety and access control to said documents using fireproof file cabinets with combination/key locks, access control to these cabinets, etc. This also limits the logical way of access, i.e., you cannot look up data if you are not physically in the same place where the data are located. Electronic tools enable us to have much simpler storage, complete traceability regarding the person responsible for creating, changing, reviewing, or validating each data, and virtually unlimited online accessibility using a convenient system of permits per user, identifying each user with their own password-protected access.
At BioClever, we have been working for years with one of the most powerful tools in the market: Clinsight, which complies with 21 CFR part 11 of the FDA and EMA standards and is certified by ISO 9001:2015. This is a highly versatile, intuitive tool with absolute traceability that allows to remotely monitor as well as manage a study, increasing the quality of data collection and review, and providing updated information on the status of the study at all times, which translates into a more efficient, transparent and reliable process with completely traceable and complete data. It is also one of the few tools that allow to export databases directly to SAS without having to export/import in Excel or another format.
Clinsight allows us to work with maximum accuracy, have each study with its own, much more personalized design and characteristics adapted to the client, but with the added value provided by a tool developed by a company of international renown that has been approved and validated by the main regulatory agencies. It offers a global solution for the development of a study, such as IWRS or SAE reconciliation, and allows to send SAEs at the same time as an investigator reports them. There is an increasing demand for these tools, highlighting the imperative need for even stricter data management processes; that is why, at BioClever, our entire Data Management team has been trained and certified to work pursuant to the guidelines of the CDISC (Clinical Data Interchange Standards Consortium).
What is the CDISC?
CDISC is a non-profit organization that has created standards to support the collection, exchange, and reporting of data and metadata in clinical research. This allows to improve interoperability between information systems to connect data from different clinical areas. CDISC was born from the need to reach a consensus on data collection and storage, so regulatory agencies always receive databases in the same way. One example of pre-CDISC standardization is the use of MedDRA for coding medical conditions.Thus, using CDISC, we can obtain large databases in which we can connect several studies or trials to each other, which also helps us to perform new inter-study data analyses or search new observations that may not have been the original study objective. By using the CDISC methodology, you can standardize formats, both during the design of the database and for any subsequent statistical analyses.
In conclusion, we would like to highlight that the software that BioClever uses is 100% compliant with these standards; that is why, BioClever is currently and seamlessly developing its databases with CDISC for any studies that request it.
We appreciate you taking the time to read this article and hope it was of your interest; however, should you have any questions or comments you would like to share with us, please feel free to contact our professionals.